If you haven’t been paying attention, medical psychedelics are on the rise, with the new industry gaining an impressive foothold even before legalizations occur. In this new move, Wesana Health will acquire Psytech, Inc., giving the company new strength to dominate this new emerging market.
The world changes quickly. Not only is Wesana Health about to acquire PsyTech, Inc., signaling even more growth in the medical psychedelics industry, but our favorite psychedelic – cannabis – has more options for consumers than ever. Take Delta 10, THCV, THC-O or Delta-8 THC for example. This half-brother of delta-9 THC provides users with a slightly less intense psychoactive high, doesn’t produce anxiety, and leaves users clear-headed and energetic. This is amazing for anyone who wants a different option. Check out our array of delta-8 THC products – along with tons of other compounds offered, and take advantage of new drug technologies and formulations.
Subscribe to the Psychedelics Weekly newsletter below:
Wesana will acquire Psytech, Inc., what will this mean?
Wesana Health Holdings, is a life sciences company that specializes in developing and delivering therapies for neurological health issues. The Chicago-based company looks to help patients overcome the damage of physical brain trauma which results in neurological, psychological, and mental health problems. The company was founded recently, in 2020, and looks to develop therapeutic solutions using psychedelic therapies including drugs like: Ketamine (and esketamine, which is currently legal), mescaline, MDMA, and psilocybin.
Psychedelitech, Inc. (PsyTech, Inc.) is a company specializing in the medical psychedelics industry, which provides clinical tools and education, as well as clinical care. The company promotes psychedelic-assisted therapy, novel methods of care and the tools to go along with them, and integrative ways for mental healthcare delivery. The company focuses a lot on the use of psilocybin therapies. The company has three parts: Tovana Solutions – a SaaS platform, Tovana Clinics – which provides a psychiatric care network, and PsyTech Connect – a community for psychedelic practitioners.
It was announced on June 13th, 2021, that Wesana Health would acquire PsyTech for $21 million, making PsyTech a completely owned subsidiary of Wesana. This will give Wesana access to all three parts of PsyTech. Wesana is looking to expand its efforts into neurological healthcare. According to CEO Daniel Carcillo (who is also a former NHL hockey player and two-time winner of the Stanley Cup), Wesana is working on new treatments and medications to treat traumatic brain injuries. He made this statement about the acquisition:
“The acquisition of PsyTech will greatly accelerate our ability to understand, analyze and improve neurological health and performance by providing a data platform on which to build our technical strategy, clinics in which to apply and accelerate our neuroscience research and relationships with many thousands of the practitioners who will leverage our medicines, diagnostics, and technology to heal people.”
The three arms of PsyTech
PsyTech has three components that Wesana will be taking over. Tovana Clinics – soon to be Wesana Clinics is a chain of mental health clinics which specialize in the delivery of psychedelic-based care, which currently involves esketamine therapy (as this is the only currently legalized psychedelic medication), and looks to incorporate new compounds as they become legal. The chain currently involves two locations, with a third set to open later this year, and about 12 more in the works that should be operational by this time next year.
PsyTech’s Tovana Solutions platform provides data collection, tracking in real-time, patient management, and general analysis tools. It also provides healthcare professionals the ability to learn current protocols and track effectiveness. The platform will be renamed Wesana Solutions.
The last arm, PsyTech Connect, is a network of over 8,000 professionals who tune in to find out about best clinical practices and protocols. Besides the network of practitioners, it also provides conferences, and educational material. The idea for Wesana is to integrate with psychiatrists across the US to expand the company and its therapeutic model.
Wesana founder and Executive Chairman, Chad Bronstein, reminds: “There are over 50,000 psychiatrists and 15,000 psychiatric practices in North America alone who will require solutions to adopt the novel and effective psychedelic-assisted therapies that already exist and are currently in development.”
Both the boards of Wesana and PsyTech have approved the acquisition unanimously. In order for it to officially go through, 2/3 of PsyTech’s shareholders must also approve. With 67% of shareholders already signed onto an agreement of support for the measure, there shouldn’t be anything getting in the way of the acquisition happening.
What psychedelic medications are already used?
The medical psychedelic movement is massively picking up speed, even if it hasn’t quite filtered through to mainstream media just yet. There are, by the way, reasons that news of this industry’s growth hasn’t made major headlines in major publications. As of right now, there are a lot of smaller biotech companies like Wesana and PsyTech getting in on it, and that means competition for the major pharmaceutical companies, which so far do have the only legal offering. Until large pharmaceutical companies can fully profit off the industry, I expect it will be kept quiet, despite major growth.
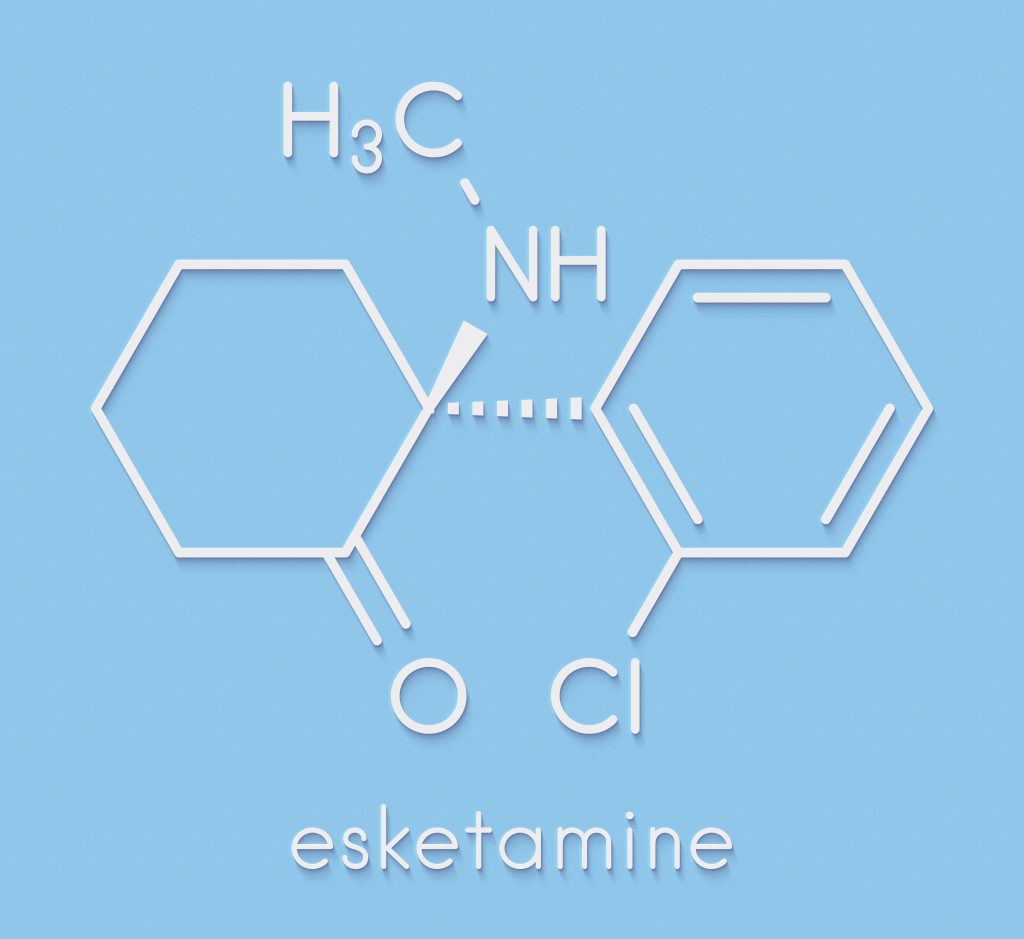
So what is currently legal? Only one medication is out called esketamine. What is this compound? Esketamine, as the name implies, is a close relative of the dissociative and psychedelic party drug, (and animal tranquiller and human anesthetic), ketamine. In 2019 the FDA approved esketamine for treatment for major depression.
In 2020, the FDA updated the approval to cover prescription for suicidal thoughts as well because of how fast-acting the compound is. Esketamine is the first new medication approval for depression which does not fit the standard model of antidepressants, as its not an SSRI, tricyclic antidepressant, or MAO inhibitor. In fact, it entirely goes against the current model for the treatment of mental illness, meaning it does not work with monoamines.
Esketamine is sold under the name Spravato, being marketed by Johnson & Johnson’s Janssen Pharmaceutical Companies. It has been approved as an anesthetic under other trade names like ketanest. Esketamine is a Schedule III substance in the US.
What psychedelic medications are on the way?
Obviously, if one psychedelic drug has been approved, which already breaks with the idea that all psychedelics are illegal (obviously not the case), then why shouldn’t it be expected that more are on the way? In fact, they most certainly are, and to show how clear it is these legalizations are coming, the US government is actually pushing for them through its own Food & Drug Administration. In fact, the two compounds its currently pushing, are specifically Schedule I drugs at the moment, but will not be for much longer. Here’s why:
In 2017, the FDA earmarked the drug MDMA as a ‘breakthrough therapy’ for the treatment of PTSD. What does this term mean? According to the FDA, “A breakthrough therapy designation is for a drug that treats a serious or life-threatening condition and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement on a clinically significant endpoint(s) over available therapies.” This designation isn’t blindly made, but generally comes at the request of a drug company, which is currently doing research that shows the compound is more promising than current options.
This description is meant to quicken research and get products to market faster. What this means, is that the FDA is pushing for a Schedule I substance – defined as a highly dangerous compound with no therapeutic value, to be on pharmacy shelves sooner, rather than later. To make it even more clear, the Multidisciplinary Association for Psychedelic Studies (MAPS) – which is the organization that won the designation for its research, is currently in phase 3 trials for an MDMA drug, which were put together in conjunction with the FDA to ensure the trials and outcomes would be in line with FDA regulation. Is there a better way to say the US government wants this drug out to consumers?
The thing is, MDMA isn’t the only drug being backhandedly pushed by the US government. In 2019, the FDA gave two separate ‘breakthrough therapy’ titles to psilocybin from magic mushrooms, for use with major depressive disorder. The first granting of this designation was given to Compass Pathways, which looks to treat the most severe treatment-resistant depression, and the second time around it went to Usona Institute, which has ongoing trials to test the efficacy of just one dose of psilocybin to treat major depression.
Conclusion
That Wesana Health is about to acquire PsyTech, is just another indication of the growing magnitude of this new industry. The acquisition also highlights not only the growing appeal of psychedelic compounds to treat mental illness, but of the networks now being put together, which will set up the entire framework of how these therapeutic services will run.
Hello and welcome! You’ve arrived at CBDtesters.co, your #1 spot for all the most thought provoking, and relevant cannabis-related news globally. Take a read-thru of the site daily to stay abreast of what’s happening in the exciting universe of legal cannabis and medical psychedelics, and sign up for our newsletter, so you never miss a thing.
Disclaimer: Hi, I’m a researcher and writer. I’m not a doctor, lawyer, or businessperson. All information in my articles is sourced and referenced, and all opinions stated are mine. I am not giving anyone advise, and though I am more than happy to discuss topics, should someone have a further question or concern, they should seek guidance from a relevant professional.







